Daily coffee doesn’t just sharpen your morning, it quietly reshapes your gut from the first sip. It triggers the gastrocolic reflex within minutes, nudges gastrin release, and leaves a measurable microbial fingerprint, including a striking surge in Lawsonibacter asaccharolyticus, in regular drinkers.
The mechanism runs deeper than caffeine. Polyphenols and soluble fiber act as prebiotics, fueling short-chain fatty acid production and dampening inflammation. Even decaf delivers the same shift, meaning the cup itself is the medicine, not just what’s in it.
Coffee’s Rapid Gut Response Starts a Chain Reaction
Your morning cup doesn’t wait around. Within minutes of that first sip, coffee activates a cascade of digestive signals (gastrin release, hydrochloric acid secretion, and bile flow) that together push your gut into gear before you’ve even finished your mug. That’s not a coincidence or a caffeine jolt: it’s your gut responding to coffee as if a meal just arrived.
Here’s how the wiring works. Your stomach and colon are connected by a reflex loop (the gastrocolic reflex) that’s designed to make room when food comes in. Normally, eating triggers it. Coffee, it turns out, pulls the same trigger, even though it’s mostly liquid. The stomach senses something’s arrived, signals the colon to contract, and suddenly you’re looking for the nearest bathroom.
How coffee activates the gastrocolic reflex
The gastrocolic reflex is a fast, hardwired response: when the stomach stretches or detects incoming food, it sends a signal down the line telling the colon to start moving. Coffee activates this reflex almost immediately, and the speed is what makes it so striking.
Part of what makes coffee so effective here is that it doesn’t just stretch the stomach, it chemically primes the whole system. Coffee stimulates gastrin release, a hormone that tells the stomach to ramp up hydrochloric acid secretion. That acid environment kickstarts protein breakdown and signals the gallbladder to release bile, which handles fat digestion further down the tract. The entire upper digestive system shifts into active mode.
The downstream effect hits the colon fast. Study by Boekema et al., authors of a narrative review on coffee’s effects on the gastrointestinal tract published in Nutrients, put it plainly:
“Distal colonic motility increases as rapidly as 4 min after coffee ingestion. These effects are unlikely to be mediated by caffeine; instead, an indirect action on the colon mediated by neural mechanisms or gastrointestinal hormones has been suspected.”
Four minutes. That’s not a slow metabolic process: that’s a reflex. And because it’s so fast, many gastroenterologists advise pairing coffee with food. A meal buffer gives the stomach something substantial to work with, which smooths out the motility spike and makes the whole transition easier on the gut lining.
About 29% of people feel this effect strongly
Not everyone sprints to the bathroom after their morning cup, but a significant share do. Research suggests roughly 29% of coffee drinkers experience a strong, compelling urge to defecate shortly after drinking. That’s nearly one in three people, a consistent pattern, not an outlier response.
What’s telling is that this defecation response shows up with decaffeinated coffee too. Caffeine independence here is a real finding: the effect persists even when the caffeine is removed, which means something else in the coffee (its acids, its chlorogenic compounds, its other bioactive components) is doing the work. Caffeine may add some intensity, but it’s not the root cause.
This is actually where a practical habit comes in that a lot of experienced coffee drinkers land on through trial and error: eating before your first cup. The logic isn’t just comfort: a meal in the stomach modulates the motility spike, giving the gut a more gradual on-ramp. It also sets up a better environment for coffee’s prebiotic compounds to interact with the gut lining, rather than hitting an empty system and triggering an abrupt response. The gut benefits are still there. The urgency is just dialed back.
Regular coffee drinkers carry a distinct microbial fingerprint
Regular coffee drinkers carry a distinct microbial fingerprint, dominated by a striking surge in Lawsonibacter asaccharolyticus, a gut bacterium that appears in dramatically higher concentrations in people who drink coffee than in those who don’t. Think of it less like coffee growing a new microbe and more like it flipping a light switch for one that was already waiting. The bacterium is there in non-drinkers too, but it’s sitting quiet, almost dormant, until coffee arrives.
Coverage by Sprudge.com co-founder of ZOE and researcher Dr. Tim Spector, lead researcher on the Nature Microbiology study behind this finding, put it plainly:
“This study highlights just how fussy our microbes can be. This Lawsonibacter microbe hangs around in suspended animation, just waiting for a cup of coffee in order to flourish.”
That image (suspended animation) is worth sitting with. It tells us something important: the microbial shift isn’t random noise: it’s a specific, repeatable response to a specific input.
How strong is the evidence? The numbers are hard to dismiss. Across a primary multi-cohort analysis of roughly 22,000 people, Lawsonibacter asaccharolyticus showed up 4.5 to 8 times more abundantly in regular coffee drinkers than in non-drinkers, with statistical significance at q < 0.001: that’s a tight threshold that accounts for the false-discovery risk you get when you’re testing hundreds of species at once. The pattern held across a broader validation pool of over 54,000 participants drawn from 211 cohorts worldwide.
The table below lays out how that fingerprint looks across different study populations, so you can see both the consistency and the gaps in the data:
| Cohort | Sample Size | Coffee Group vs. Control | Lawsonibacter Abundance | Other Notable Shifts | Statistical Significance |
|---|---|---|---|---|---|
| Multi-cohort (USA/UK primary) | ~22,000 | Regular drinkers vs. non-drinkers | 4.5–8× higher in drinkers | 115 species positively associated (e.g., Faecalibacterium, Alistipes) | q < 0.001 |
| Integrated public cohorts | ~54,198 (211 cohorts) | Regular drinkers vs. non-drinkers | 4.5–8× higher in drinkers | Not specified | Not specified |
| Food survey cohort | ~23,000 | Never / moderate / heavy drinkers | Absent in never-drinkers; ~4× higher in moderate/heavy | Distinct profiles across all three categories | Not specified |
| Decaf vs. caffeinated comparison | Subset (size unspecified) | Decaf vs. caffeinated vs. none | Higher in both drinker groups, independent of caffeine | Not specified | Not specified |
| Rural vs. urban cohorts | 74 cohorts (52 urban, 20 rural) | Urban Western vs. non-Western rural | Median 75% prevalence in urban; median 2.4% in rural | Not specified | Not specified |
But here’s where we have to pump the brakes. Every number in that table comes from observational, cross-sectional data. That means researchers looked at who drinks coffee and measured what lives in their gut: they didn’t randomly assign one group to drink coffee for six months and then compare. So while the association between coffee consumption and Lawsonibacter asaccharolyticus is consistent and statistically strong, no randomized or mechanistic study has yet confirmed that coffee causes this shift. It’s possible that people who drink coffee also eat, sleep, or live differently in ways that independently shape their microbiome.
There’s a second claim floating around in coverage of this research: that the microbial fingerprint is distinctive enough to predict coffee drinker status with “high accuracy.” That’s a compelling idea, but the performance metrics that would let us evaluate it (things like AUC, sensitivity, and specificity) haven’t been published in the primary literature. Without those numbers, “high accuracy” is a marketing phrase, not a scientific finding.
None of this makes the data less interesting. It makes it more interesting, actually: because the consistency across 54,000 people and 211 cohorts is the kind of signal that earns a proper mechanistic investigation. What we’re looking at right now is a very clear “what:” the “why” is still being worked out.
Coffee’s Polyphenols Feed Your Gut Microbes Directly
Fermentable polyphenols in coffee reach your colon largely intact, where gut bacteria break them down into metabolites that fuel microbial growth and reshape the community living there. Most people think of coffee as a caffeine delivery system. The prebiotic chemistry happening downstream of that first sip is a different story entirely, and it runs on compounds most coffee drinkers have never heard of.
The two main substrates doing this work are soluble fiber and chlorogenic acid. They arrive in your large intestine undigested, and that’s exactly the point.
How much soluble fiber and polyphenols are in coffee?
Soluble fiber in a standard filtered cup delivers roughly 1.5 grams of prebiotic substrate: not a trace amount, but a meaningful dose that gut microbes can actually work with. That number comes from a peer-reviewed nutritional analysis in Nutrients, which also mapped the conversion chain that follows.
The dominant polyphenol is chlorogenic acid (CGA). Chemically, CGA is an ester: meaning it’s two molecules bonded together: caffeic acid and quinic acid. When gut bacteria hydrolyze CGA, they cleave that bond and free the quinic acid. Quinic acid then gets converted further into hippurate, a metabolite your kidneys eventually excrete in urine.
That conversion chain (chlorogenic acid → quinic acid → hippurate) isn’t just metabolic housekeeping: it’s the feeding pathway for Lawsonibacter asaccharolyticus, the same bacterium we saw surge in coffee drinkers in the previous section. Multi-omic analysis in that same Nutrients study confirmed the link directly: elevated quinic-acid-derived metabolites in the bloodstream correlated with higher populations of L. asaccharolyticus and other beneficial taxa. The bacterium isn’t just present in coffee drinkers: it’s there because of this specific chemical pathway.
So the soluble fiber provides bulk fermentation substrate, and the polyphenol cascade provides a selective growth signal. Together, they don’t just feed the microbiome broadly, they steer it.
Coffee metabolites drive SCFA production and gut barrier strength
SCFA production, specifically the generation of butyrate, is where the microbial activity in your colon translates into something your body’s cells can feel directly. Short-chain fatty acids are the end products of bacterial fermentation. When microbes like Faecalibacterium and Bifidobacterium longum break down the fiber and polyphenol substrates from coffee, they produce SCFAs as metabolic byproducts.
Literature review on coffee and microbiota put it plainly:
“Coffee intake can mediate changes in microbiota through caffeine and/or other coffee components or metabolites like micronutrients and phenolic compounds… A study in a rodent model found that long-term coffee consumption led to an increase in short chain fatty acids (SCFAs) producing bacteria such as Faecalibacterium and Bifidobacterium longum.”
Butyrate is the SCFA your colon cells prefer above all others. Colonocytes, the cells lining your large intestine, use butyrate as their primary fuel source, the same way your muscles use glucose. When butyrate supply is adequate, those cells maintain tight junctions: the molecular “seals” between them that prevent undigested particles and bacterial fragments from leaking into the bloodstream. This is the mechanism behind the phrase “gut barrier integrity,” and it’s why butyrate production gets so much attention in gut health research.
Coffee also contains melanoidins, the dark, high-molecular-weight compounds formed during roasting when sugars and amino acids react under heat. Melanoidins reach the colon largely intact, where they act as antioxidants and appear to dampen inflammatory signaling in the gut lining. Combined with the anti-inflammatory effects of polyphenols themselves, this gives coffee a secondary layer of gut protection beyond just feeding beneficial bacteria.
Here’s where it’s worth slowing down, though (because the story gets more complicated than “more bacteria, better health”).
The assumption that greater microbiome diversity automatically means better health outcomes is one of the most repeated ideas in gut health content, and it doesn’t hold up cleanly under scrutiny. Diversity is a proxy measure, not a health outcome. L. asaccharolyticus, for all its consistent presence in coffee drinkers, hasn’t yet been linked to specific, measurable health benefits in human trials. And a diversity increase isn’t inherently positive if the taxa rising alongside the beneficial ones include opportunistic or pathogenic strains.
What matters isn’t the diversity number: it’s the composition and the metabolic activity of the community. Butyrate production from Faecalibacterium and Bifidobacterium longum is a concrete functional output. That’s a more meaningful signal than a diversity index going up.
To see what measuring that SCFA output actually looks like at the lab level, this video walks through a real assay predicting personalized SCFA production profiles from gut microbiota, the same kind of methodology researchers use to connect coffee compounds to microbial metabolite output:
The biochemical pathway from your morning cup to butyrate production in your colon is real and reasonably well-mapped. The open question (whether decaf coffee drives the same pathway, since caffeine isn’t the actor here) is exactly where we go next.
Decaffeinated Coffee Drives the Same Microbiome Shift
Decaffeinated coffee delivers its core microbiome benefits through three non-caffeine compounds: polyphenols, N-methylpyridinium, and soluble fiber, each of which feeds or signals your gut bacteria through a distinct pathway. Strip out the caffeine, and the machinery that reshapes your microbial community is still fully intact. That’s the part most people miss when they assume caffeine is doing the heavy lifting.
Think of caffeine as the horn on a car: loud, noticeable, easy to credit for the whole experience, but the engine running underneath it, the one actually moving you forward, is something else entirely.
Here’s how the map breaks down across those three compounds:
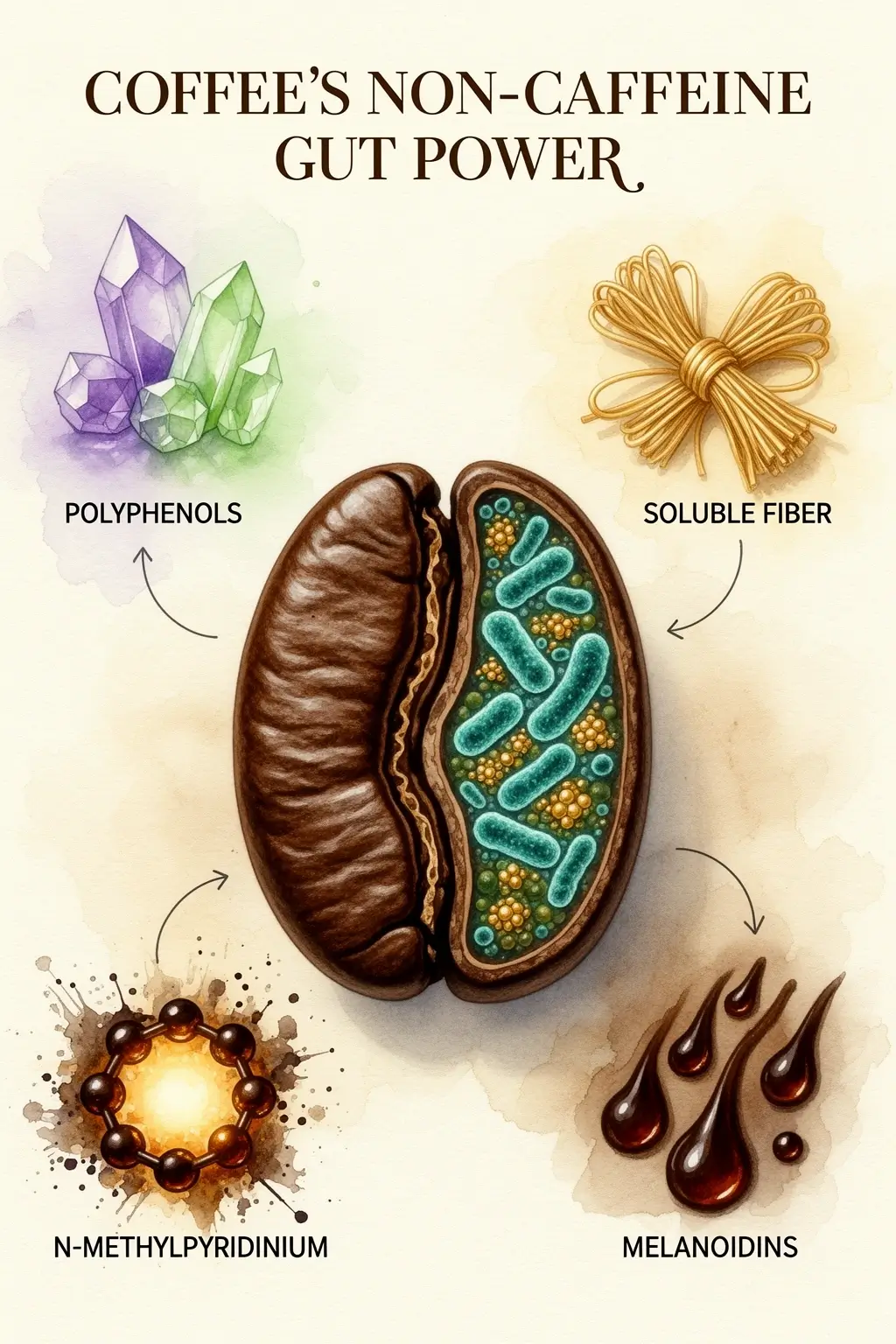
Polyphenols are the most abundant active agents in both caffeinated and decaffeinated coffee. They’re largely indigestible in the small intestine, which means they arrive in the colon intact, right where your microbiome lives. Once there, specific bacterial strains metabolize them and, in exchange, produce short-chain fatty acids and other anti-inflammatory byproducts. The bacteria aren’t just eating the polyphenols; they’re being selected by them. Strains that can break down chlorogenic acid and other coffee phenolics gain a competitive edge, which is a big part of why Lawsonibacter asaccharolyticus blooms so reliably in regular coffee drinkers.
N-methylpyridinium (NMP) forms during roasting: it’s a heat-driven transformation product, not something present in the raw bean. NMP has been shown to reduce stomach acid secretion and appears to modulate gut barrier function. Darker roasts carry more of it, which is one reason some people with sensitive stomachs actually tolerate dark roast better than light, counterintuitive as that sounds.
Soluble fiber in coffee acts as a straightforward prebiotic. It’s fermentable material that feeds your existing microbial population and nudges the community toward strains associated with better metabolic and inflammatory outcomes.
None of these three compounds disappear when caffeine is removed. The decaffeination process targets caffeine specifically (a small alkaloid molecule) and leaves the polyphenol matrix, the fiber content, and the roast-derived compounds like NMP essentially untouched.
The research backs this up directly. Researchers behind a landmark study published in Nature Microbiology found that:
“The microbiome associations showed substantial independence from caffeine for the observed impact.”
That’s not a minor footnote. It means the microbial fingerprint we’ve been attributing to “coffee” is really a fingerprint of coffee’s non-caffeine chemistry. Caffeine may accelerate gut motility in the short term, that’s a separate, well-established mechanism, but the deeper structural changes to your microbial community appear to run on a different fuel entirely.
For anyone avoiding caffeine for sleep, anxiety, or cardiovascular reasons, this is genuinely good news. The gut-health case for decaffeinated coffee is not a consolation prize: it’s the same bet, just quieter.
Acid Reflux, FODMAPs, and How Much Coffee Is Actually Safe
Coffee’s well-documented acidity and its ability to relax the lower esophageal sphincter can provoke uncomfortable GERD symptoms, but only in people who are already susceptible. For most healthy adults, the research keeps coming back to the same reassuring answer: moderate intake doesn’t meaningfully raise the risk of serious upper-GI trouble. The irritation that some people feel is real, but it’s not universal, and the mechanism behind it is more specific than “coffee is acidic.”
How coffee relaxes the lower esophageal sphincter
Esophageal sphincter relaxation is the actual mechanical event that turns a cup of coffee into a source of reflux for vulnerable drinkers. The LES is a ring of muscle that acts like a one-way valve between your esophagus and stomach, one it lets food down and keeps acid from traveling back up. Coffee contains compounds, including caffeine and certain chlorogenic acids, that signal that muscle to loosen its grip. When it does, stomach acid has an easier path upward, and that’s when you feel the burn.
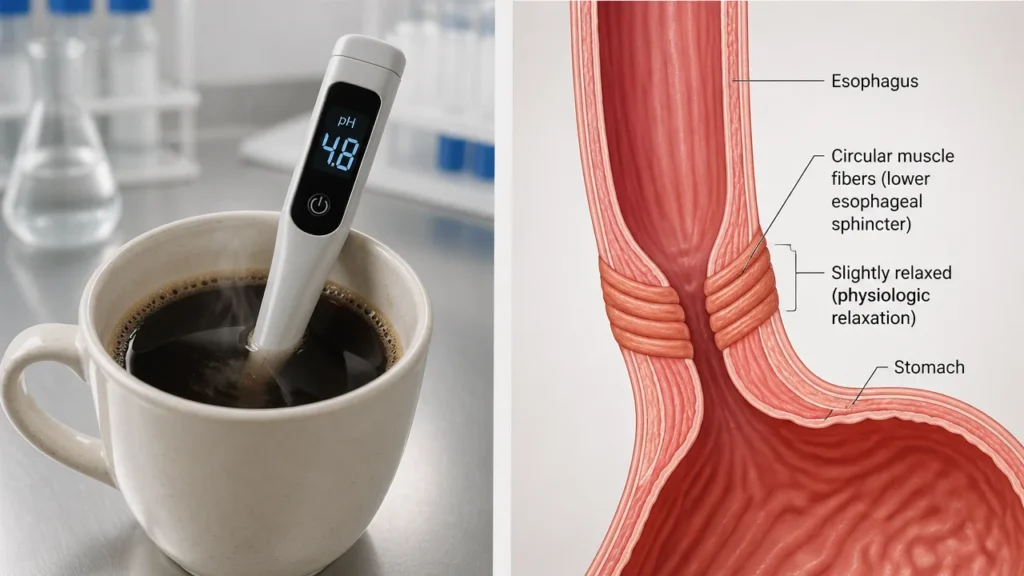
Here’s what matters, though: that relaxation response is dose-dependent and highly individual. Large-scale reviews examining hundreds of thousands of participants consistently find no significant link between moderate coffee consumption and major upper-GI disorders in healthy people. The LES relaxation happens, but a healthy, well-toned sphincter recovers quickly. The people who feel it most are those whose sphincter tone is already compromised (think chronic GERD sufferers, people with hiatal hernias, or anyone who’s eating large meals right before drinking).
So the acidity of coffee, which typically sits around pH 4.5 to 5, is less of a villain than its reputation suggests: black coffee is actually less acidic than orange juice. The real trigger is the sphincter signal, not the pH number on a meter.
FODMAP thresholds and timing strategies for coffee
FODMAP classification for coffee is something most people have never seen laid out clearly, and that missing information is exactly why so many sensitive drinkers either avoid coffee entirely or drink it without any strategy and pay for it later. Monash University, the institution that developed and maintains the FODMAP framework, has actually tested coffee and given it a tiered rating based on serving size.
Here’s how the Monash FODMAP thresholds break down for instant coffee specifically:
- Green (low-FODMAP, generally safe): 1 teaspoon / 4 g — the standard serving for a single cup
- Amber (moderate, proceed with caution): 150 g — well beyond normal use
- Red (high-FODMAP, likely problematic): 1 cup measured by weight / 189 g — again, far outside typical consumption
What this tells us is that a normally brewed cup of coffee, made with a standard teaspoon of grounds or instant, sits comfortably in the green zone for most people following a low-FODMAP approach. The FODMAP load only becomes a real issue when consumption climbs well past what most people would drink in a sitting.
That said, FODMAPs aren’t the only variable in play. A 2023 meta-analysis of 122,000 participants found that 34.9% of coffee drinkers reported GERD symptoms, compared to 30.7% of non-drinkers, a real but modest gap that tells us coffee raises risk for some people without being the primary cause for most. The difference between the two groups likely comes down to individual susceptibility, not coffee consumption alone.
Timing is where you get the most practical leverage: drinking coffee on an empty stomach removes the buffering effect that food provides: stomach contents dilute acid and slow gastric emptying, which reduces the pressure pushing against the LES. A small meal or even a snack before your first cup makes a measurable difference for people who are on the sensitive side. Smaller servings spread across the morning work better than a large cup all at once, for the same reason.
The practical takeaway: if coffee causes you discomfort, the answer probably isn’t elimination, it’s calibration. Portion size, timing relative to meals, and your own baseline LES health are the three levers worth adjusting before you give up the cup entirely.
Building a Gut-Friendly Coffee Routine
A well-timed gut-friendly coffee routine pulls together meal timing, roast selection, organic beans, and sweetener guidelines into one coherent daily practice (that might sound like a lot of variables, but each one traces back to the same underlying logic we’ve been building across this entire piece: coffee’s benefits flow from its polyphenols and fiber, and your job is to keep those compounds working for your gut instead of against it). Get the context right, and coffee stops being a gamble and starts being a reliable part of how you feel every day.
The four levers below aren’t arbitrary rules: each one addresses a specific friction point between coffee’s chemistry and your digestive system.
Meal timing and roast selection work together.
Smart meal timing pairs with medium-to-dark roast selection to give you coffee’s prebiotic upside without the acid irritation that undermines it. Here’s the mechanical reason: drinking coffee on an empty stomach sends a concentrated acid load directly into your gut lining, which can spike gastric acid production and, in sensitive people, accelerate motility to the point of discomfort rather than benefit. Eating first (even something small with protein and fat) buffers that acid load and slows gastric emptying just enough for the polyphenols to do their prebiotic work.
Roast choice matters here more than most people realize. Medium-to-dark roasts contain higher concentrations of N-methylpyridinium (NMP), a compound that actually suppresses stomach acid production. Lighter roasts taste brighter partly because they carry more chlorogenic acids, the same acids that, in large amounts, can tip sensitive stomachs toward reflux. If you’ve been blaming coffee for your heartburn and you’re drinking a light roast on an empty stomach, you’ve stacked two acid-amplifying choices on top of each other.
By aligning these two habits: eating before you brew and choosing a roast your stomach can work with: you turn coffee into a reliable gut-health ally rather than a coin flip.
Organic beans protect the microbiome you’re trying to build.
Organic and biodynamic beans limit the pesticide and mycotoxin load that can quietly disrupt the microbial balance coffee is simultaneously trying to improve. Think about the irony: you’re drinking coffee specifically because its polyphenols feed beneficial bacteria like Lawsonibacter asaccharolyticus, but if the beans carry residual pesticides, you’re delivering a microbial disruptor in the same cup. It’s like fertilizing a garden and spraying herbicide at the same time.
Biodynamic beans take this a step further: they’re grown under stricter soil-health standards, which tends to produce beans with a richer phytochemical profile. Whether that translates to a measurable gut benefit over standard organic is still an open research question, but the floor is the same: fewer compounds that work against your microbiome.
If caffeine is your specific problem: it triggers reflux or keeps you wired past midnight: decaf is a straight swap, not a consolation prize. The microbiome benefits we’ve traced throughout this piece come from the polyphenols and soluble fiber, not the caffeine. Decaf preserves both. Your Lawsonibacter population doesn’t know the difference.
Sweetener guidelines close the last gap in your daily routine.
Carefully chosen sweetener guidelines, anchored by unsweetened oat or almond milk, protect the microbial balance that everything else in this routine is designed to build. High-sugar creamers feed the wrong bacteria, the opportunistic strains that thrive on simple sugars and crowd out the beneficial populations your coffee polyphenols are cultivating. Artificial sweeteners create a different problem: compounds like sucralose and saccharin have been shown in multiple studies to alter gut microbial composition, and not in a favorable direction. You’d be spending all that effort on roast selection and meal timing, then undermining it with what you pour in the cup.
Unsweetened oat milk is a reasonable middle ground, it adds a small amount of beta-glucan fiber, which is itself prebiotic, without the sugar spike or the artificial chemistry. Almond milk works too, though it contributes less fiber.
Here’s a simple daily checklist to hold it all together:
- Eat first: Have protein and fat before your first cup (even a handful of nuts counts).
- Choose your roast: Medium-to-dark for acid sensitivity; light roast only if your stomach handles it well.
- Go organic when you can: Prioritize it over single-origin marketing if you have to choose.
- Consider decaf after noon: Caffeine’s motility effect compounds over the day, decaf keeps the prebiotic benefits running without the stimulant load.
- Watch what goes in the cup: Skip the flavored creamers and artificial sweeteners. Use unsweetened oat or almond milk, or drink it black.
- Keep portions honest: One to two cups per sitting, not a continuous drip. Your gut needs time to process each dose before the next one arrives.
None of this requires a complete overhaul of how you drink coffee. It’s mostly about sequencing what you already do: eating before you brew, reaching for a darker roast, and reading the label on your creamer: small adjustments, but they’re the ones that actually move the needle.
Real Talk: What Most People Miss About Coffee’s Gut Game
Q: Why doesn’t coffee cause the poop effect in everyone?
A: About 29% feel it strongly due to individual sensitivity in the gastrocolic reflex wiring, but decaf works the same way showing it’s not caffeine—it’s the acids and compounds hitting your colon in 4 minutes flat. Everyone’s gut responds differently based on baseline motility, so don’t expect universal bathroom runs.
Q: Can Lawsonibacter asaccharolyticus levels really predict if you’re a coffee drinker?
A: No, that’s hype—the observational data across 54,000 people shows 4.5-8x higher levels in drinkers with strong stats, but no published AUC or accuracy metrics exist yet. Cross-sectional studies prove association, not prediction power; causality needs RCTs.
Q: Is more microbiome diversity from coffee always a good thing?
A: Nope, diversity is just a proxy—coffee boosts Lawsonibacter and others, but without human trials linking it to outcomes, and if opportunists rise too, it’s neutral at best. Focus on outputs like butyrate from Faecalibacterium, that’s the real health signal.
Q: What if you’re on low-FODMAP—can you still drink coffee daily?
A: Yes, standard 1 tsp instant or grounds is green zone per Monash, only huge volumes like 189g hit red. Pair with food to buffer, since empty-stomach timing amps symptoms more than FODMAP load itself for most folks.
Q: Why do dark roasts sometimes feel better on sensitive stomachs than light?
A: Dark roasts pack more N-methylpyridinium from roasting, which cuts stomach acid secretion—light roasts keep higher chlorogenic acids that irritate. Eat first regardless, but roast choice flips the acid dynamic without losing prebiotic punch.
Q: Does decaf fully match regular coffee for gut benefits if caffeine bugs you?
A: Absolutely, microbiome shifts like Lawsonibacter surge come from polyphenols, fiber, and NMP—all intact post-decaf processing. Nature Microbiology confirms independence from caffeine; it’s quieter motility but same structural microbial wins.





